The risk in having too many risks…
Confusing failure modes for risks is one of the most common structural mistakes in medical device risk analysis — and one of the most costly to fix later. This article explains the difference between hazards, hazardous situations, and harm under ISO 14971, why a bloated risk analysis undermines your whole risk management process, and how one simple syntax rule can help you build a cleaner, more actionable document from the start.
Do you have more than 40 risks in your device risk analysis — and it's not even invasive?
Most likely, they are not risks. They are failure modes. And confusing the two is one of the most common — and costly — mistakes I see in early-stage medtech.
A risk list that has grown out of control creates real problems:
It dilutes focus away from the risks that actually matter — the ones you should be able to recite off the top of your head. It opens the door to inconsistencies and duplication in a document so large that no colleague will review it in detail, but that an auditor will flag immediately. It turns every product feature into a hazard or risk control, which then warrants stricter testing requirements down the line. And it makes traceability in post-market surveillance and clinical evaluation a genuine operational nightmare.
I've seen many well-meaning startups suffer through the consequences of a badly designed risk analysis. The QARA who built it might feel proud of its thoroughness. But the rest of the team loses interest and never truly owns their risk areas. Management stops using it for decision-making. Product design becomes cluttered with risk controls — warnings, untouchable features — that nobody can explain.
This kills the collaborative and iterative spirit that is essential for good risk management.
So what's the difference between a risk and a failure mode?
A risk analysis table is built from three distinct layers, as described in ISO 14971 and ISO 24971:
Hazard categories — the nature of the potential harm (energy, software, misuse — full list in ISO 24971)
Hazardous situations — the circumstances in which people are exposed to a hazard, including failure modes and external causes
Harm — the actual injury or damage to health that may result
The most common mistake is conflating hazards with hazardous situations — that is, treating failure modes as if they were risks in their own right. The terminology doesn't help, admittedly.
One simple strategy to keep your risk analysis clean
Use a fixed syntax to write your risks consistently. Here's one I find practical:
THERE IS A RISK OF [who] [hazard type faced] ORIGINATING FROM [list of failures and hazardous situations] WHICH MAY LEAD TO [harm type — pick only the highest level]
Two examples:
For a hardware device: There is a risk of the patient coming into contact with high voltage (electrical energy), originating from a) damage to the connecting cable, b) manufacturing defect, c) poorly designed insulation — which may lead to electric shock.
For a SaMD: There is a risk of the physician receiving inaccurate output from the device (incorrect medical decision), originating from a) algorithm design limitations, b) algorithm execution error, c) user interface failure, d) cybersecurity attack, e) unclear instructions for use — which may lead to delay in treatment.
Notice how multiple failure modes collapse into a single, well-defined risk. That's the point. Your risk analysis becomes shorter, more focused, and far easier to maintain over time.
If you're building the table manually, write the syntax in your header row. If you're using AI-assisted tools, enter it as a prompt constraint or use it to validate the output. If you're reviewing an existing table, run each row against it.
A risk analysis should be accessible to the whole team, actionable in decision-making, and sustainable as the product evolves. Getting the structure right from the start is one of the highest-leverage things a QARA can do in an early-stage company.
Deep Dive: Getting the Structure Right
Risk Analysis vs FMEA
Both are part of the Risk Management process under ISO 14971, but they serve different purposes and are not interchangeable.
Risk Analysis is mandatory. It is the top-level document that captures your device's safety profile — the full picture of what could go wrong, for whom, and with what consequences. Think of it as the billboard for your device's safety. It needs to tell a meaningful story, not overwhelm the reader with noise.
FMEA (Failure Mode and Effects Analysis) is a supporting analytical method — good practice, and often expected by auditors, but not explicitly required by ISO 14971 as a named technique. It is the drill-down tool: you take each component, subsystem, or process and ask systematically, how could this fail, and what would the effect be?
The same FMEA logic appears under different names depending on the domain:
In SaMD, it is often formalised as a Software Hazard Analysis (required under IEC 62304 as part of software risk management)
In usability engineering, it underpins the Use-Related Risk Analysis (URRA), which traces use errors and abnormal use to potential harm — a core deliverable under IEC 62366-1
In cybersecurity, it is effectively a vulnerability analysis or threat modelling exercise (with reference to MDCG 2019-16 and IMDRF guidance on cybersecurity)
Each of these domain-specific analyses follows the same logic: identify how something could fail, then trace that failure to a potential harm. The outputs of all of them feed into one Risk Analysis for your product — not multiple separate risk documents.
This is where the structural confusion often starts. Teams run an FMEA, a URRA, and a software hazard analysis, and then copy the failure modes directly into the Risk Analysis table. The result is a document that mixes hazards, hazardous situations, and failure modes in the same column, under the label "risk." Multiply that across a product with many subsystems, and you quickly reach 60, 80, or 100+ rows — most of which are not risks at all.
The three-layer structure
ISO 14971 and its companion standard ISO 24971 are clear on the terminology, even if teams frequently blur the distinctions in practice:
Hazard: a potential source of harm — an inherent property of the device or its environment (e.g. electrical energy, ionising radiation, software decision output)
Hazardous situation: the circumstance in which a person is exposed to a hazard — this is where failure modes, use errors, and external conditions live
Harm: the physical injury or damage to health or property that results
A well-structured risk analysis row moves through all three layers. The failure modes — however many there are — belong in the hazardous situation column, not in a row of their own. That single structural choice is what keeps the document manageable.
A note on harm classification
For the harm column, the IMDRF Adverse Event Terminology provides a standardised, hierarchical coding system that is increasingly expected in technical documentation and is directly useful in post-market surveillance reporting. Using it consistently from the start — rather than free-text descriptions — saves significant effort later when feeding into your PMSR or PSUR.
Practical checklist
Can every row in your table be read using the [who / hazard type / originating from / harm] syntax? If not, it may be a failure mode, not a risk.
Are failure modes consolidated under their parent risk, rather than listed as standalone rows?
Is the harm column using consistent, ideally IMDRF-aligned terminology?
Could a new team member read the Risk Analysis and understand the device's core safety story in under 30 minutes?
References
ISO 14971:2019 and ISO 24971:2022 — available at a significantly lower cost than ISO directly via the Estonian Standards store (legitimate national standards body, same official text)
IMDRF Adverse Event Terminology browser — for standardised harm classification
IEC 62304 (software lifecycle) and IEC 62366-1 (usability engineering) — for domain-specific hazard analysis requirements that feed into the Risk Analysis
Methodology note: This article is based on two original LinkedIn posts (first, second) written by me, reflecting my professional experience and personal perspectives on risk management in medical device development. Claude AI assisted in combining and expanding the posts into a broader article for this blog, integrating background context, regulatory references, and a structured Deep Dive section. All regulatory perspectives and practical recommendations are my own, and all content has been reviewed by me for accuracy.
The next strategic frontiers of medtech
Where should medtech and femtech companies focus their international expansion efforts after establishing EU and US market presence? This article analyses eight high-potential markets across four global regions — Anglosphere, East Asia, MENA, and Latin America — ranked against three strategic criteria: ROI and medtech infrastructure, femtech readiness, and regulatory alignment with international harmonisation frameworks including MDSAP and CE Mark mutual recognition agreements. Whether you are mapping your next regulatory strategy or building a market entry roadmap, this evidence-based analysis offers a practical starting point — with an honest look at what the data says and where gut instinct still has a role to play.
After my popular recent poll, which strategic markets deserve to be your and my next medtech focus beyond US-EU? Note: beyond, not instead of!
What are the factors we want to optimise?
ROI & MEDTECH INFRASTRUCTURE: markets that offer the highest investment and monetization potential, with advanced payer structures and tech scaling capabilities.
FEMTECH READINESS: regions where the cultural and policy discourse is opening to women’s health, where clinical research is advanced, and femtech innovation and startup ecosystem are thriving.
REGULATORY ALIGNMENT: countries that align with international medtech harmonisation efforts, for example being members of MDSAP or with mutual recognition agreements (MRA) towards CE mark and FDA.
With the help of Claude, I ran an advanced analysis to find the right countries for these categories. More details on the results summarised below.
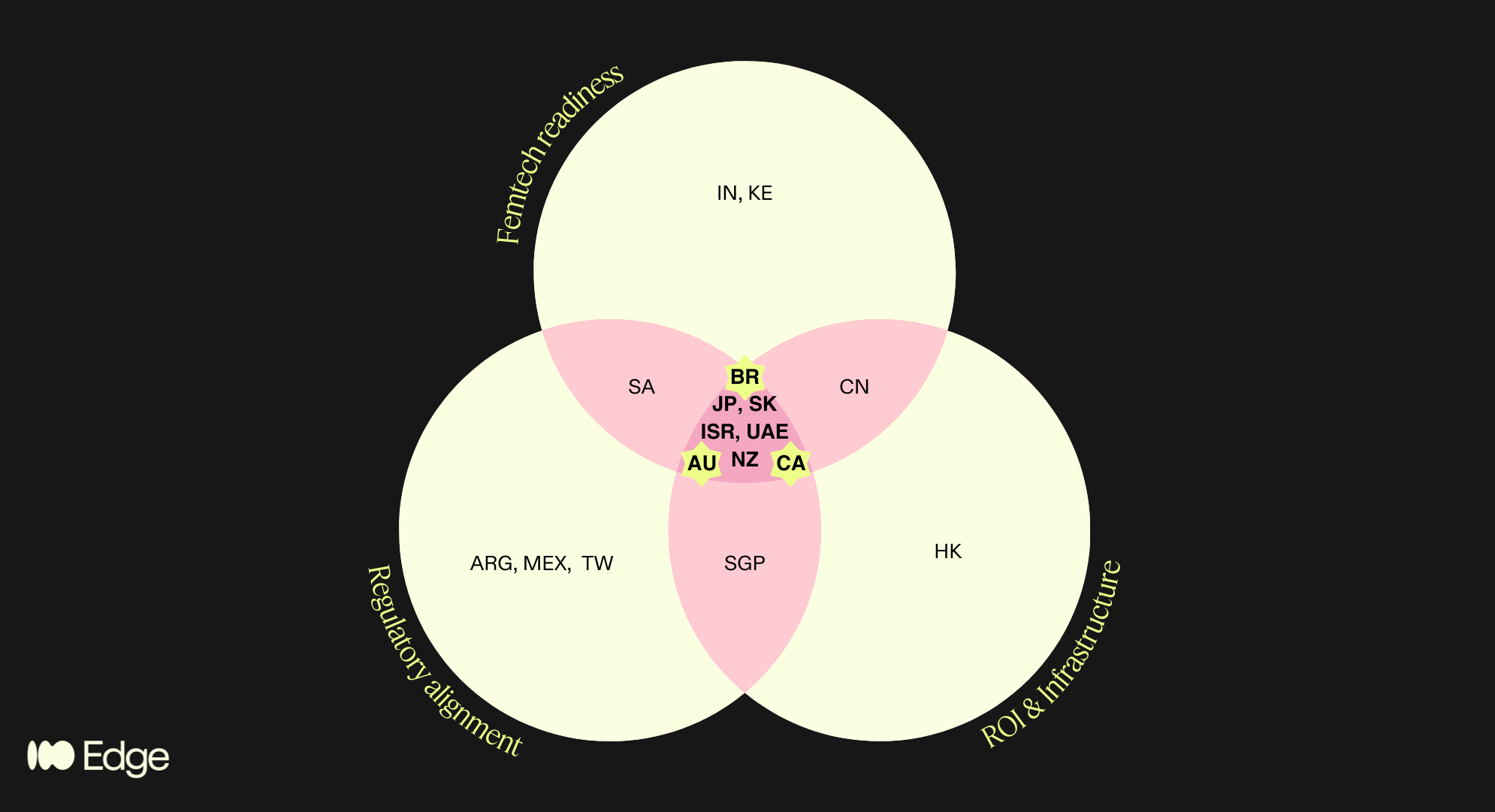
Regions that satisfied multiple criteria are highlighted in the pink areas of the diagram. This brings us to 8 high potential regions, spanning the 4 geographies of my poll!
Anglosphere: Canada, Australia, New Zealand
“BRIC”+East Asia: Japan, South Korea
MENA: Israel, UAE
LatAm: Brazil
What does it mean for your market strategy? If you are in femtech and looking to expand after EU/US proof-of-concept - AND not planning to exit directly - then these are your markets. Save and share the post, and narrow down the list based on further criteria relevant to you and your business.
To me and my business, I applied the following additional criteria:
Accessibility: where can my cultural roots and the languages I speak help me?
Market size: >20M for highest chances of leads and indirect product reach for my clients
Heterogeneity: the more diverse the better for clinical research relevance
Geopolitical stability: low risk of conflicts and market volatility
Which takes us to the winners!
🇧🇷 Brasil: as half-Brazilian I’ll be proud and excited to explore the femtech, clinical and regulatory space and build connections there! DM me to discuss!
🇦🇺 🇨🇦 Australia and Canada: it’s a tie! I already work with these markets, no big lift, but good pointer to go a bit deeper. Spoiler: more MDSAP content coming, like it or not..!
🇦🇪 UAE: Although it didn’t pass my filters, something in my gut tells me not to give up on it. I’d love to partner up with medtech experts and providers in that area.
This aligns well with the results from the poll: 52 votes, indicating predominant interest in "anglosphere" (32%) and "emerging" (30%), incl. LatAm.
Thanks to all who voted, commented and even called me to share their passionate insights!
Deep dive
The framework: three lenses, not one
We structured the analysis around three criteria, chosen because they reflect the real bottlenecks that founders face when entering new markets.
Regulatory alignment (MDSAP / MRAs). For a CE-marked or FDA-cleared device, the most efficient path into a new market is one that recognises or aligns with those approvals. The Medical Device Single Audit Program (MDSAP) is the primary international harmonisation vehicle, currently comprising the US (FDA), Canada (Health Canada), Brazil (ANVISA), Australia (TGA), and Japan (PMDA). Beyond MDSAP, a growing network of Mutual Recognition Agreements (MRAs) extends this regulatory compatibility further — most notably, the EU–Australia MRA, Israel's deep alignment with EU MDR, South Korea's MFDS harmonisation pathway, and GCC's increasing adoption of CE and FDA as reference standards.
Femtech readiness. This goes beyond market size. It asks whether the cultural, policy, and clinical infrastructure of a market is genuinely open to women's health innovation — or whether femtech companies will spend the first two years of market entry fighting the problem rather than solving it. We looked at startup density, clinical research activity, regulatory openness to digital health, and the status of women's health policy discourse in each candidate country.
ROI and medtech infrastructure. Commercial viability ultimately depends on whether a market can pay for solutions and scale them. This means evaluating healthcare financing structures (private payer vs. public reimbursement vs. out-of-pocket), digital health infrastructure, startup ecosystem depth, and the presence of credible distribution and partnership networks.
Only markets that scored meaningfully across all three lenses made the final shortlist. That intersection — regulatory alignment AND femtech readiness AND commercial infrastructure — is the filter that separated 8 markets from the 25+ we considered.
The 25-country universe and how it narrowed
We mapped approximately 25 countries across the three criteria, drawing on data from:
Institute for Economics & Peace Global Peace Index 2024 (163 countries) for geopolitical stability scoring
EF English Proficiency Index 2024 (116 countries) for accessibility
UN Population data 2024 for market size
World Bank ethnic fractionalization data for demographic heterogeneity (relevant to clinical research representativeness)
Vestbee, Dealroom, Speedinvest, Grand View Research, Startups Magazine for femtech market data by country
MDSAP member country documentation and publicly available MRA registers for regulatory alignment
Countries in only one or two circles of the Venn include genuinely interesting markets — Singapore (strong MDSAP alignment and infrastructure but too small for standalone commercial focus), India (exceptional femtech growth projected at 17.8% CAGR to 2030 per Grand View Research, but regulatory alignment is a work in progress), South Africa (emerging femtech ecosystem with strong policy momentum, but infrastructure gaps and geopolitical risk), China (significant commercial potential but regulatory and geopolitical friction makes it a poor fit for EU-anchored companies), and Kenya (one of Africa's most active femtech ecosystems, but not yet operationally viable for most European companies without a dedicated local strategy).
These are not dismissed markets — they are markets for a different risk appetite or a later stage.
The 8 that made the intersection
These are the markets that sat inside all three criteria simultaneously. For each, we break down the case across the three categories.
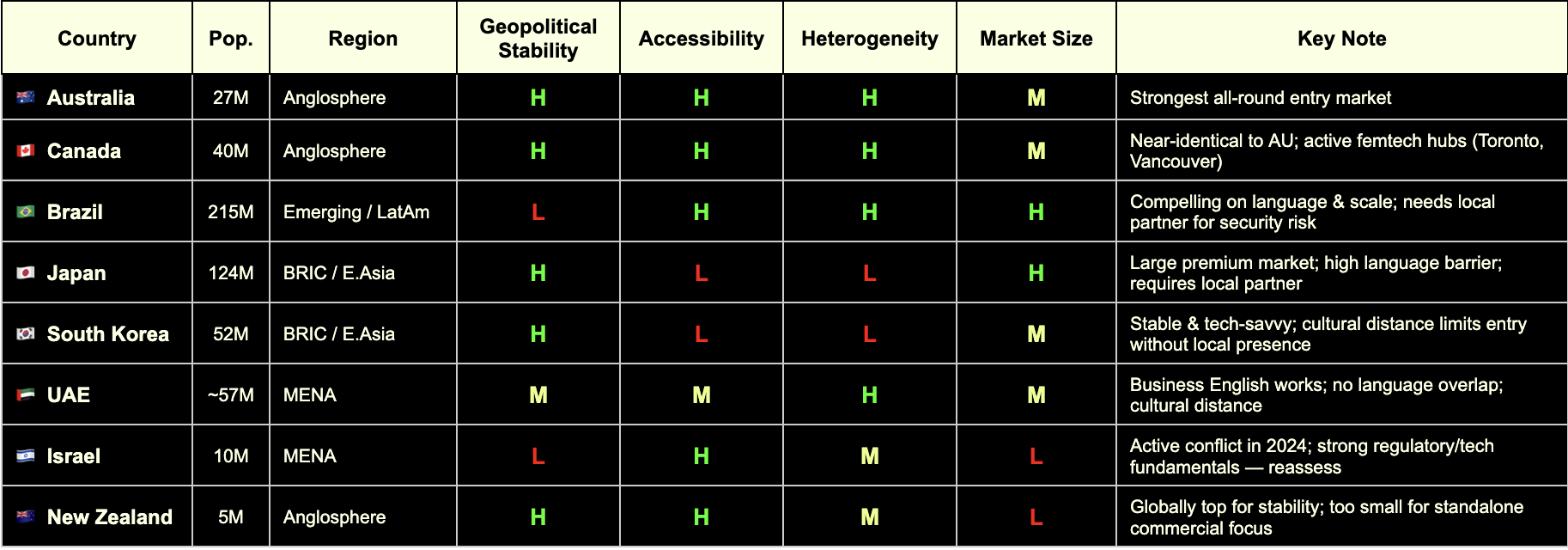
🇦🇺 Australia
Regulatory Alignment
MDSAP full member (TGA)
EU–Australia Mutual Recognition Agreement in place — CE mark directly supports TGA approval pathway
Strong alignment with ISO 13485 and IEC 62304 for software-based devices
Femtech Readiness
Mature digital health market with active government investment (Australian Digital Health Agency)
Women's health policy discourse is open and progressive; no significant cultural friction for femtech categories
Active medtech and healthtech startup ecosystem, particularly in Melbourne and Sydney
ROI & Infrastructure
Population ~27 million; 27% foreign-born (one of OECD's highest) — valuable for diverse clinical evidence
Mixed public/private payer system; growing private digital health reimbursement
English-speaking, Commonwealth-aligned, low operational complexity
GPI 2024: #17 — high geopolitical stability
🇨🇦 Canada
Regulatory Alignment
MDSAP founding member (Health Canada)
A single MDSAP audit covers Canada alongside the US, Brazil, Australia, and Japan — significant efficiency gain
Health Canada increasingly aligned with FDA on digital health device classification
Femtech Readiness
Toronto and Vancouver are active femtech hubs; Femtech Canada is an established national ecosystem
Strong clinical research infrastructure with diverse patient populations
Progressive women's health policy environment; high awareness and destigmatisation of femtech categories
ROI & Infrastructure
Population ~40 million; ~27% immigrant share — comparable diversity to Australia
Robust private and public payer landscape; provincial variation requires navigation
English and French markets; English operations are broadly sufficient for market entry
GPI 2024: #13 — highly stable
🇧🇷 Brazil
Regulatory Alignment
MDSAP full member via ANVISA — the most underutilised structural advantage for European companies entering LatAm
A single quality audit unlocks market access across all five MDSAP jurisdictions simultaneously
ANVISA has been modernising its device framework; registration timelines have improved significantly
Femtech Readiness
Growing digital health and femtech ecosystem, particularly in São Paulo and Rio de Janeiro
Large, young, highly digitally engaged female population
Significant unmet need in reproductive health, maternal health, and menstrual care — high receptivity to femtech solutions
ROI & Infrastructure
Population ~215 million — the largest in Latin America by a substantial margin
Highest ethnic fractionalization score globally (World Bank) — clinical data generated here is maximally representative
⚠️ GPI 2024: #131 — operational complexity is real; security considerations, contract risk, and local partner dependency must be factored in
Local partner infrastructure is a prerequisite, not an option
🇯🇵 Japan
Regulatory Alignment
MDSAP full member via PMDA (Pharmaceuticals and Medical Devices Agency)
PMDA is one of the most rigorous regulatory bodies globally; MDSAP audit significantly reduces duplicative burden
Regulatory pathway for digital health devices (SaMD) is well-defined
Femtech Readiness
One of the world's most advanced femtech markets by per-capita spend on menstrual health and reproductive technology
Strong clinical research infrastructure and data quality
⚠️ Cultural norms around women's health are evolving but remain conservative in some categories — market education investment required
ROI & Infrastructure
Population ~124 million; high healthcare spending and premium consumer health culture
High-margin market for validated, evidence-backed products
⚠️ EF English Proficiency Index: #92 out of 116 — significant language barrier; local partner or Japanese-speaking regulatory lead is non-negotiable
GPI 2024: #11 — very high stability
🇰🇷 South Korea
Regulatory Alignment
MFDS (Ministry of Food and Drug Safety) has progressively aligned with FDA and EU MDR frameworks
MDSAP compatibility pathway is expanding; not yet full membership but convergence is directional
Government actively promoting K-healthcare internationally — creating reciprocal openings for foreign market entry
Femtech Readiness
High digital health adoption and smartphone penetration; strong consumer appetite for health tracking and wearables
Growing awareness of women's health, particularly in fertility and menstrual health tech
Active startup ecosystem with increasing women's health focus
ROI & Infrastructure
Population ~52 million; advanced payer structures and tech scaling infrastructure
Government investment in digital health innovation is substantial
⚠️ EF EPI: #50 — business English limited outside Seoul; local presence required
GPI 2024: #46 — stable, with standard geopolitical considerations given peninsula context
🇮🇱 Israel
Regulatory Alignment
Israeli MOH operates with strong alignment to EU MDR and FDA standards — one of the most harmonised non-MDSAP markets globally
CE mark is a recognised reference standard; dual EU/FDA submission strategies are well-established
Deep bilateral ties with both EU and US regulatory bodies
Femtech Readiness
Exceptionally strong clinical research infrastructure; Israel produces a disproportionate number of medtech innovations relative to population
Highly developed VC and medtech ecosystem; femtech companies including HeraMed are globally recognised
High awareness and openness to women's health innovation; destigmatisation is advanced relative to regional peers
ROI & Infrastructure
Population ~10 million — small absolute market, but high income and high health spend per capita
English proficiency high (EF EPI: #51); business environment is highly accessible to European operators
Strong potential as a clinical trial site and R&D partnership hub, regardless of commercial scale
⚠️ GPI 2024: #155 — reflects active conflict situation as of the 2024 index; market entry decisions require live geopolitical risk assessment, not a static one. The underlying regulatory, clinical, and commercial infrastructure remains among the strongest in this cohort
🇦🇪 GCC (UAE & Saudi Arabia)
Regulatory Alignment
Neither is a formal MDSAP member, but UAE and KSA have adopted CE mark and FDA clearance as primary reference pathways for device registration
UAE MOHAP and Saudi SFDA increasingly streamlined; CE mark provides a strong starting point
GCC Standardization Organization (GSO) harmonisation creates some regional regulatory efficiency
Femtech Readiness
Saudi Arabia's Vision 2030 explicitly references women's empowerment and healthcare improvement — femtech is policy-aligned
UAE has the most open and internationally diverse women's health market in the region
Growing investor interest in femtech and digital health across the Gulf; GITEX Health and Arab Health are active deal-making forums
ROI & Infrastructure
UAE: ~89% of resident population is expatriate (UN data) — genuinely diverse patient population and cosmopolitan business environment
English is the functional operating language for business in both markets
Premium private-pay market; reimbursement structures favour high-value, evidence-backed products
⚠️ GPI 2024: UAE #53 (improved 31 places in 2024 — single largest improvement in the index); KSA requires ongoing monitoring
Cultural distance to women's health topics exists in some categories — local partnership and community navigation are important
🇳🇿 New Zealand
Regulatory Alignment
MDSAP full member (Medsafe)
Strong regulatory alignment with Australia (TGA); joint Trans-Tasman regulatory considerations apply
Simple, transparent regulatory environment — among the most accessible globally for device registration
Femtech Readiness
Progressive women's health policy environment; high health literacy and digital engagement
Maori and Pacific women's health is an active policy and research priority — relevant for inclusive femtech design and clinical evidence
English-speaking, low cultural friction across all standard femtech categories
ROI & Infrastructure
Population ~5 million — too small for standalone commercial focus
Best positioned as a clinical trial jurisdiction, regulatory proof-of-concept market, or Australasian stepping stone alongside Australia
GPI 2024: #4 — among the most geopolitically stable countries on earth
High income per capita; public system (Pharmac) for reimbursement is conservative but private market is accessible
What this means for women’s health startups
The eight markets are not interchangeable, and we are not suggesting a company should pursue all of them simultaneously. The practical read is this:
For a femtech company at the EU/US proof-of-concept stage, with limited expansion resources and no existing presence outside Europe, Australia and Canada are the lowest-friction first moves. They are MDSAP members, English-speaking, culturally accessible, and have enough market depth to justify the investment.
Brazil becomes compelling the moment you have a local partner or are prepared to build one. The MDSAP pathway is a structural advantage that is consistently underutilised by European companies, and a population of 215 million — with the highest demographic diversity of any market on this list — is a significant clinical research and commercial prize.
Japan and South Korea are high-reward markets that require deliberate localisation investment. The regulatory frameworks are accessible through MDSAP and MRA alignment; the human infrastructure needs to be built.
Israel and the GCC are specialist considerations — Israel for companies with strong clinical trial and research ambitions, the GCC for companies targeting the premium private-pay women's health segment and building into the Middle East and North Africa corridor.
New Zealand is genuinely valuable as a regulatory and clinical site — less so as a primary commercial target.
The 4th lens…
At Edge Compliance we want to keep serving US and EU market entries (EU as a continent, incl. UK and CH).
What this analysis taught us is where to put our next focus in terms of partnerships, network, learning, advocacy.
In order to avoid spreading ourselves too thinly, we applied some filters to narrow down the list of high potential regions to a smaller selection. The Edge Compliance lens considers:
Cultural accessibility
Market size
Demographic heterogeneity
Geopolitical stability
Criteria for market selection for Edge Compliance. Which are relevant to your business?
This analysis led us to choose the following areas of focus in addition to our existing focus and expertise:
🇧🇷 Brasil
🇦🇺 Australia
🇨🇦 Canada
🇦🇪 UAE
We trust this will help us help YOU with future-proof regulatory strategy and foresight that is ahead of trends.
References
Institute for Economics & Peace GPI 2024
EF English Proficiency Index 2024
UN World Population Prospects 2024
World Bank Ethnic Fractionalization Data
Vestbee Femtech Market Overview (2024)
Dealroom Femtech Report (2024)
Speedinvest Femtech Investment Analysis
Grand View Research India Femtech Market Outlook 2024–2030
MD+DI FemTech Analytics (2024)
Startups Magazine Southeast Asia Femtech Report
The Recursive CEE Femtech Analysis (2025)
Methodology note: This Deep Dive is based on my original LinkedIn post, reflecting my professional experiences and personal perspectives. Claude AI assisted in elaborating the topic into a broader article by integrating personal notes, literature research, fact-checking and deeper insights on the topic. All analysis and regulatory perspectives are my own, and all content has been reviewed by me for accuracy.
PFAS: wait-and-see Vs precautionary principle
When data is limited, do you default to safety or wait for proof of harm? This post explores the widening regulatory gap between the US and EU regarding PFAS - the "Forever Chemicals."
If you had to decide whether something is safe based on limited data, which way would you default?
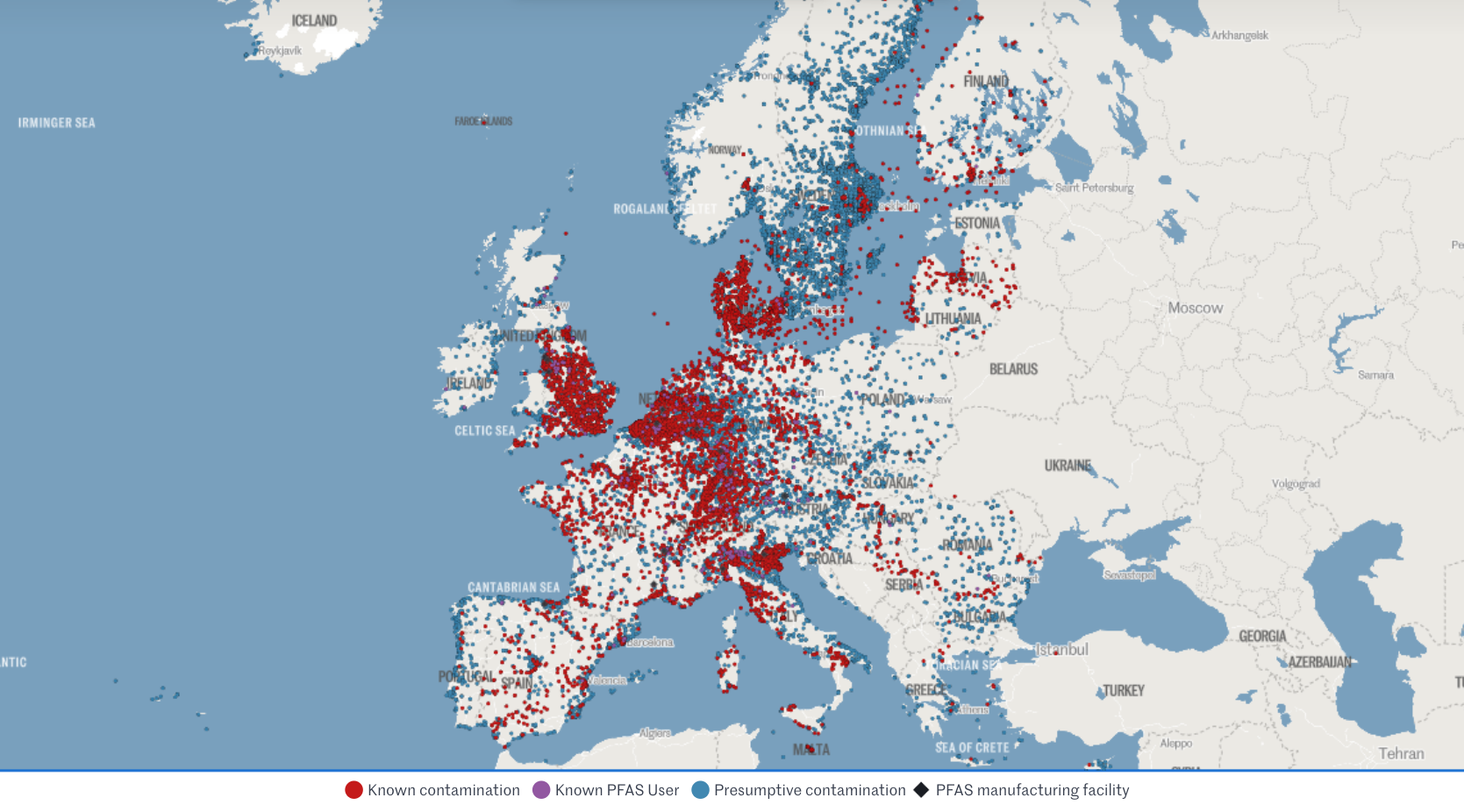
Let's look at recent regulatory developments re "Teflon-like" chemicals (PFAS) in cosmetics and medical devices. Per- and polyfluoroalkyl substances (PFAS) are highly inert synthetic chemicals which makes them sought after for both everyday uses and specialist ones. However, they are so inert that biology cannot break them down. They persist in the environment and accumulate in creatures at the top of the food chain: us.
The regulatory approach to PFAS, also called Forever Chemicals, is another staggering example of the US vs. EU cultural divide.
U.S. wait-and-see approach
🇺🇸 Context: In 2024, FDA launched the Modernization of Cosmetics Regulation Act (MoCRA) which required registration of all cosmetics and listing of all their ingredients. This allowed FDA a fresh overview on PFAS' use in cosmetics, which inspired recent research.
🇺🇸 Research: A December 2025 report revealed that 51 types of PFAS are intentionally used in 1,744 cosmetic formulations in the US, commonly in makeup and even baby products.
🇺🇸 Conclusion: Due to a lack of critical toxicological data and acute toxicity, the safety of 76% of these compounds could not be definitively established. FDA deemed current evidence insufficient to justify a federal ban, opting instead for continued monitoring.
🇺🇸 Note: The FDA excluded environmental considerations and the assessment of unintentional degradation products, which are often the most harmful (e.g., PFOA and PFOS).
EU precautionary principle
🇪🇺 Context: The EU is already phasing out PFAS over concerns regarding long-term health effects and environmental contamination.
🇪🇺 Research: Rising concentrations in water streams and human blood (even in teenagers) are increasingly suspected to suppress the immune system and increase risks of cancer, infertility, thyroid dysfunction, and metabolic dysregulation.
🇪🇺 Conclusion: Action and monitoring stepped up at national and union level.
> This month, France has banned PFAS in all cosmetics (as well as clothing textiles and ski waxes).
> Yesterday, the European Environment Agency (EEA) kicked off a mandatory EU-wide program to systematically monitor PFAS in drinking water.
> Meanwhile, European Chemicals Agency (ECHA) is evaluating a proposal to ban 10,000 PFAS as a broad category, with stricter concentration limits (ppb levels) expected by October 2026.
🇪🇺 Note: The EU had already restricted all PFAS and even banned some under the REACh and the POPs regulations (which also impact allowed limits in medical devices under MDR).
Which side would you take? Personally, I’m leaning EU on this one.
Sources:
- FDA’s report
- EEA programme
- Forever pollution project (image credits)
FDA’s new guidance on general wellness
This post critiques the widening regulatory gap between the US and EU following the FDA's new wellness guidance, highlighting how lower barriers for bold health claims in the US may sacrifice essential quality drivers and complicate global strategies for startups.
Yesterday's release by FDA on wellness vs medical device leaves me with a bitter aftertaste. Why?
I'm usually enthusiastic about policies that lower the barrier to market entry for health products. I'm less enthusiastic about those that eliminate the quality drivers from it..
My main concerns under this guidance:
> General wellness products have no QMS requirement, especially digital ones. So when the guidance says you can now display biomarkers even with some disease reference as long as "the product has validated values" for those biomarkers, it doesn't really mean anything. How do they validate? According to what? Where? Claims get bolder and accountability weaker.
> We will see more products being Class IIa medical devices in EU (with QMS auditing and device file review) while facing zero expectations in the US as general wellness.
> The gap between EU and US regulatory approach gets wider. EU released a "similar" guidance in Sep 2025 emphasising the opposite, with increased focus on mechanism of action and technology rather than relying on claims only. US heads the other way, making it all the more complicated for us RA 🥴
> It will be harder for startups to design their product and strategies for the two main western markets simultaneously. They will be pushed even heavier towards wellness-first but in my experience they get easily stuck there.
> This bold approach may be (too) specific of this administration. Will it then outlive it? It is also clearly result from the WHOOP controversy, given the number of references to Blood Pressure measuring wrist-worn devices. Pretty solid legal and lobby teams there.
One example that puzzles me in particular is the one about glucose monitoring via "minimally invasive microneedle technology" for which FDA says they will apply enforcement discretion as a low risk device. Since I'm currently working on the biocompatibility testing requirements for a device that is hand held by doctors using gloves (👀), I cannot help but finding it unfair towards the rest of the sector.
So I hope you will excuse my slightly less upbeat post this time.
I'm generally excited about the expansion of the definition and agree with the rationale of most of the examples provided.
I'm curious to see what it will mean for international harmonisation and for the opportunities it will open for my clients at this interface!
MDR/IVDR proposal for simplication
This post highlights the European Commission's groundbreaking proposal to overhaul and simplify the MDR and IVDR frameworks, promising more proportionate rules for low-risk devices, reduced administrative burdens for SMEs, and a modern, digital-first approach to medtech regulation in the EU.
12 hours ago the European Commission published THE MOST AWAITED AND CRUCIAL DEVELOPMENT IN A DECADE: its proposal for simplification of the MDR and IVDR. 👏
Alert: it is still only a proposal, albeit official, which has been submitted to the European Parliament and the Council, but will need to go through the ordinary legislative procedure to become binding Union law.
From a first diagonal read, what struck my attention:
🎉 More room for Class I devices, incl software (THANK YOU!)
🎉 Simplified interaction with AI Act
🎉 Codified instruments for open dialogue on classification and access to expert panels
🎉 Easier "equivalence" concept including use of synthetic data,
🎉 Lower NB fee structure for SMEs
🎉 Extended reporting timelines and validity of certificates
🎉 Reduced scope of surveillance audits and conformity assessment
🎉 Built-in flexibility for public health emergencies, breakthrough/orphan devices (i.e. life-threatening, rare, untreated diseases), supply-chain disruptions
Interestingly, but unsurprisingly, it proposes additional requirements for cybersecurity conformity and reporting (beyond what qualifies as medically "serious").
I will share more details of how this would impact specifically medical device startups especially in digital health and femtech.
While it is still ONLY A PROPOSAL, it is sign that EU is listening and actively working to "make [the current rules] easier, faster and more effective and further promote competitiveness, innovation and a high-level of patient safety in this key sector"
We're excited to follow the development of the legislative decision-making process and wait eagerly for the change of an era this (or its variants that will result) will bring to the European medtech sector!
PCCP beyond AI
Very exciting trend of femtech apps integrating with wearable data! How does this work for the regulated ones? I wanted to share this clever use of PCCP from Natural Cycles° from last year which impressed me.
What's PCCP?
Pre-determined Change Control Plan is a regulatory instrument devised by FDA - as a European is I'm most jealous of. It was designed to enable AI devices, which by design need to be able to evolve their accuracy in the field, getting smarter the more data they acquire. Traditionally, any change to the accuracy and performance of a device required a regulatory resubmission (still the case in EU) and up to 90 days of review wait.
With PCCP you can get pre-approval for a reasonable range of performance that you anticipate and accept.
What I found clever, is that Natural Cycles°, the pioneer of regulated fertility awareness, used PCCP not for AI changes but for variability of source data from different wearables.
While, as far as I'm aware, they currently integrate only with ŌURA and Apple Watch, this clears the way for them to swiftly add any more integrations to their conception/contraception suite as long as they fit their predefined specs (see table in pdf).
This is an example of how:
1️⃣ Regulatory instruments that are smart and abreast with the times enable even more innovation than what they primarily intended to,
2️⃣ Femtech is riding the wave of biomarkers ensuring most users can be served irrespective of which devices they choose - it's not just the iOS vs Android divide anymore!
3️⃣ Scientific research and clinical partnerships will see an incredible boost of opportunity from all this data, finally compensating for the lack of data that we know womens health has suffered until now!
What else could we use PCCP for? And until when can we have a similar toolkit in Europe under MDR? 🫠
NC's current integrations here
Link to full 510k summary here
What can we learn from… Australia?
What if the interaction with regulators was more personal?
This week, I was interviewed as part of the Therapeutic Goods Administration's research for improving health software regulation.
Since we registered a SaMD client in Australia, we were contacted to take part in a 1 hour call with ORIMA Research on the TGA's behalf. We had the chance to discuss our experience and to give suggestions on what would help other digital health companies enter the Australian market compliantly, for example:
🔍 how do companies find out whether they are regulated?
🔍 what is key for them to know in order to navigate the regs?
🔍 what's clear / unclear in the regs?
🔍 what could be attracting digital health companies to Australia?
🔍 what would put them off from doing it compliantly?
I would love to see the EU doing the same. But then, in practice, who? The European Commission? The national Competent Authorities? The Notified Bodies? Team-NB? MDCG? It gets complicated before even starting..
Yes, sometimes the EC issues calls for comments on certain regulations. The problem I have with this is a) the free-text format, which is an invite for whining and venting, and b) the lack of accountability, i.e. does anyone read it? what happens with it?
In contrast, I really appreciated:
🌟 The structured discussion format, still with some liberty to digress,
🌟 The face-to-face personal interaction, which encourages trust,
🌟 Knowing a report with clear actions will come out of it and disseminated.
Kudos to the TGA and lovely experience chatting to Jack Disher at ORIMA.
We look forward to the report!
Regulation without borders
Starting two new client projects this week, one on food supplements in France and one on in-vitro diagnostics in Germany, both in womens health!
Very few medtech consultants would feel comfortable touching other verticals (even from MDR to IVDR). But my career started like that when, honestly, I didn't have a choice! Now it's what I enjoy the most, and what I built my agency around.
The hard competences boil down to a few common traits, irrespective of sectors, regs and countries:
➡️ Regulatory definition / classification
➡️ Manufacturing requirements
➡️ Claims and label compliance
➡️ Responsible Person / Entity role
➡️ Notification / Submission procedures
➡️ Review interaction
➡️ Launch and Distribution
➡️ Post-market reporting
After all, it's all about health accountability, and humans have really one way of expecting it - the rest is often noise.
Personally, I find it super fun to come across these analogies, transfer learnings from one area to another and even anticipate cross-sector currents. Excited to get going!
Review timelines for FDA 510k clearance
How long does it take from FDA submission to clearance?
Let's look at the recent data.
The 510k database can be exported and analysed. Format is not humanly readable but makes a fun ChatGPT exercise.
Here is the result of me playing with the database from devices cleared last months (Aug and Sep 2025).
❗ The normal distribution appears to peak around 90 days, the legal obligation for FDA to respond to submissions. Around 30% of submissions were cleared within that timeframe.
❗ Nice peak at 30 days - but don't be too wishful! These are expedited reviews, e.g. changes to existing 510ks or based on prior agreements or expected updates.
❗ Less exciting peak around 270 days, i.e. 9 months. Most submissions receive an Additional Information request, which gives manufacturers 180 days to respond and restarts the clock for FDA after that (further 90 days).
Lesson here?
If you're planning a 510k, a realistic estimate for clearance is nothing less than 6 months. This is what applied to 2/3s of the 400+ applications cleared most recently.
Good quality submissions and preliminary discussions with FDA on the fundamental topics can help prevent Additional Information requests and thus increase the chances of receiving clearance within 90 days.
Does your experience confirm this too?
I will dig more into this database in the coming posts with more insights.
The WHOOP saga
WHOOP ’s current FDA row is properly binge-worthy. Material for the next Lincoln Lawyer season on Netflix?
But until then, some personal reflections on why it matters for digital health and wearables.
This season’s hottest episodes:
🎞️ Ep. 1 : WHOOP launches Blood Pressure Insights (BPI) as a Wellness feature but claiming medical grade insights.
🎞️ Ep. 2 : FDA’s surveillance picks it up and issues a Warning Letter (made public with exceptional urgency) arguing against the medical disclaimers given the “inherent association” of BP with the diagnosis of hypo/hypertension,
🎞️ Ep. 3 : WHOOP refuses to pull the feature and takes it public/political, meeting with RFK Jr and attacking FDA’s integrity on social media.
I get it, it’s tough to live on the line. Enjoying the aura of “medical-grade” without the burden is the dream of many, but it's getting harder. I’ve been there with multiple startups, and deeply empathise with some of the operational and financial challenges they faced in getting that balance right - often in absence of clear guidelines.
But now: guidance is there, WHOOP already has an FDA-cleared ECG feature (i.e. a QMS) and likely the budget... then why not route the BPI feature under their existing regulated org? Whether from the start or in response to the warning. How is taking up this massive fight a better strategy?
In smaller cases, it would ring a quality culture and integration issue. But in this one, it’s seems a fight on principle - while enjoying the extra PR of being the torch bearer for the freedom of wearables worldwide.
Meanwhile, Hilo by Aktiia quietly secures BP clearance with medical indication for its bracelet without the fuss. 👀
If you’re in the borderline medical space, this is a defining moment.
➡️ Disclaimers may be shorter-lived than ever, careful if you’re relying on those.
➡️ Not all companies are the WHOOP or SPACEX of the ton. Don’t assume this aggressive strategy would work for you, play smart yes, but sustainable. PR and legal repercussions can be devastating for fundability.
➡️ Hire QARA professionals who know how to navigate the redlines vs the negotiables of borderline products.
As Blythe Karow put it in her BEAUTIFUL long read on this story:
“The art lies in reading between the lines and addressing the specific compliance issues rather than fighting fundamental regulatory doctrine.”
Meanwhile, in real life: A friend told me "my sleep/stress score from my watch is looking weird... am I sick??". Familiar? Apparently, WHOOP had an internal policy in place during COVID that employees should stay home if their score was lower than a certain threshold - they either had the virus or could easily get it. If this is how we use these tools, what's so bad in providing assurance of quality and accuracy in the first place?
Only time will tell.. For now, pass the popcorn 🍿
EU AI Act deployment
Since August 2nd the EU AI Act is in force. But is it?
In practice: not much today, but the clock has started. If your device includes an AI component or uses AI to support decisions it’s time to take a closer look.
For high-risk systems, including many AI-based medical devices, there’s a 36-month transition to comply, i.e. phased implementation. However, some provisions apply earlier (e.g. banned uses of AI, codes of conduct).
Here’s what I see across medtech:
1. Confusion around scope and classification, e.g. AI as a tool for CSV or as part of the intended use?
2. Assumptions that MDR = AI Act compliance, thus reactive attitude to QMS updates upon NB feedback rather than in a proactive manner
3. Teams don't know how to resource it.
Good thing is that I also see a booming AI-related offering from QARA consultants and training providers which can help if you’re stuck on any of the above points. Cool examples (among many others):
• AI-first QARA frameworks and training e.g. Johner Institut GmbH https://lnkd.in/dBSuFfie,
• AI agents for compliance-checking and even FDA review outcome prediction such as Lexim AI or Acorn Compliance,
• GenAI embedded in eQMS tools such as Formwork from OpenRegulatory or Matrix One
What would help your team implementing the AI Act? Curious to hear your challenges and to help you with the right support.
Steep rise in FDA fees for 2025-2026
Alert 🫰 Steep rise in FDA fees from this October:
+19% Annual establishment registration fee from $9,280 to $11,423 (this is the one you pay every year for keeping the right to place a device on the market)
+7% Application fees, e.g. 510k submission from $24,335 to $26,067 (this is the one-off fee for review of a product submission file)
Bad news for early stage medtech businesses and SMEs, in particular since no "small business discount" nor waivers apply on the establishment fee at first registration.
Note, small businesses may qualify for waiver on the establishment fee (2nd year on) and a reduced application fee (e.g. 510k for $6,517 instead of $26,067, new fees) under the SBD programme. Conditions are based on gross sales and justification of "financial hardship", rather than on company size. Worth looking into.
See latest MDUFA fees on the FDA website at this link.